Study results
AQNEURSA was evaluated in a Phase III, multinational, randomized (1:1), double-blind, placebo-controlled, crossover trial (N=60).1,2,4
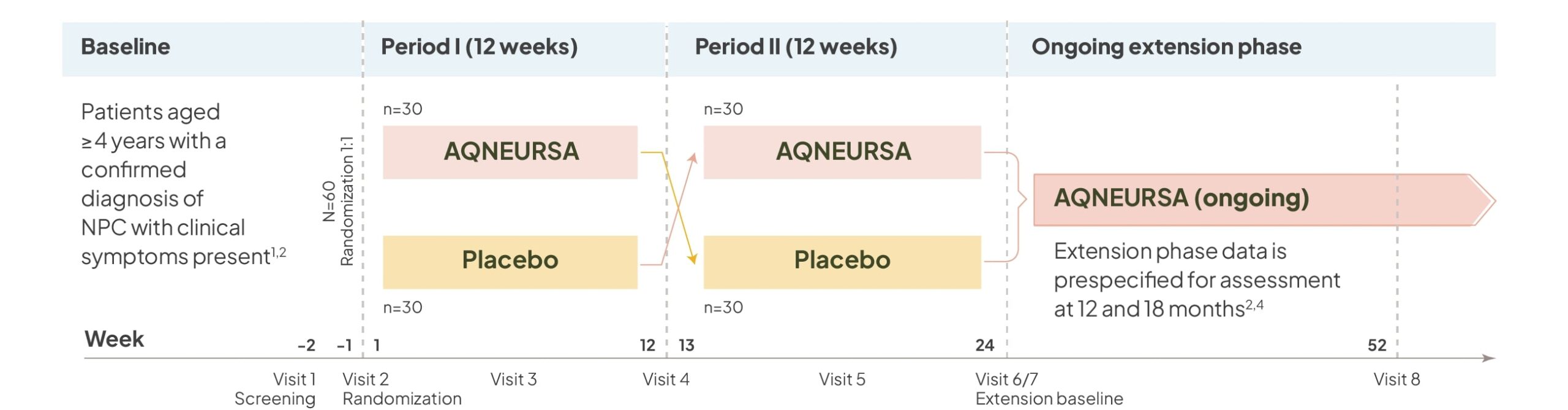
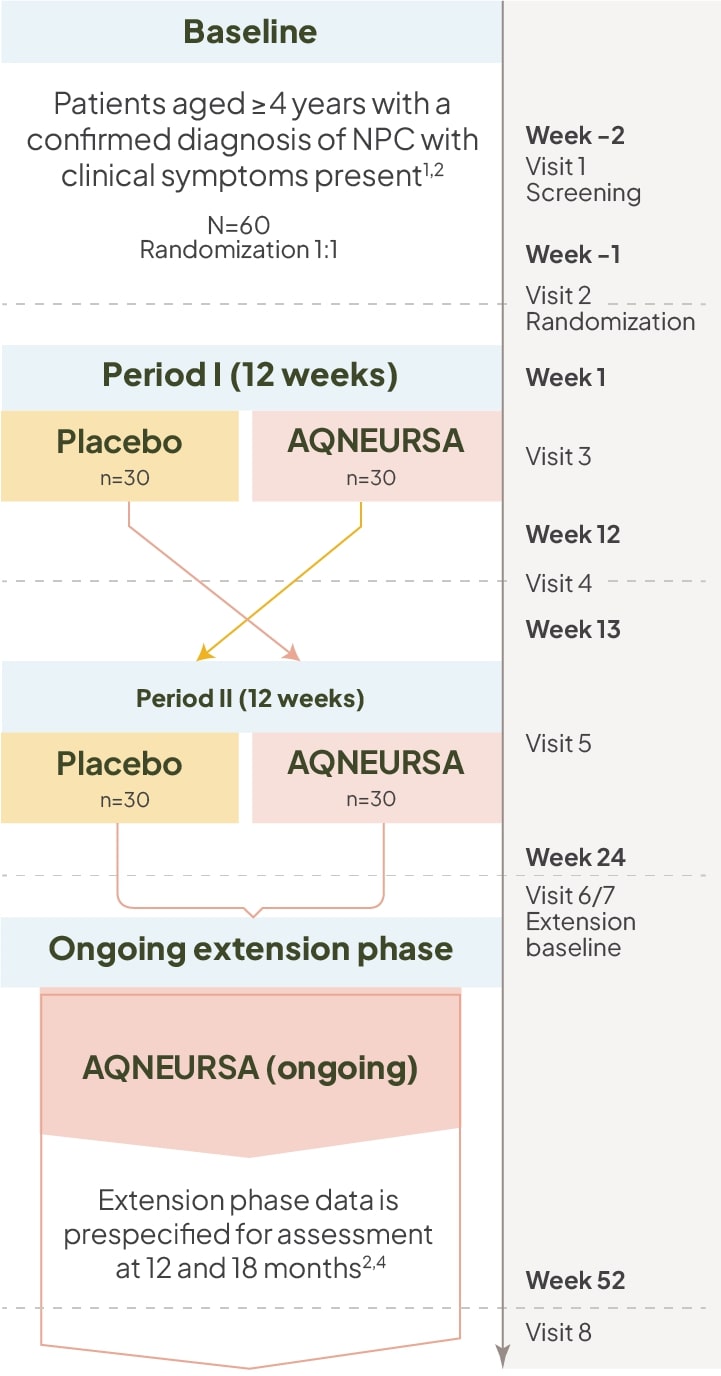
Select inclusion/exclusion criteria
| Select inclusion criteria | Select exclusion criteria |
|---|---|
| Male or female aged ≥4 years with a confirmed genetic diagnosis of NPC at the time of signing informed consent.5 | Simultaneous treatment with any investigational therapies other than miglustat (miglustat is not approved by the FDA for the treatment of NPC; arimoclomol was not approved by the FDA at the time of the study).2,5 |
| Written informed consent signed by the patient and/or their legal representative/parent/impartial witness.5 | Patients who have any known hypersensitivity or history of hypersensitivity to acetyl-leucine (DL-, L-, D-) or derivatives, excipients of the AQNEURSA sachet, or excipients of the placebo sachet.5 |
| Weight ≥15 kg at screening.5 | Current or planned pregnancy or women who are breastfeeding.5 |
|
Patients must fall within:
AND |
Patients with severe vision or hearing impairment (that is not corrected by glasses or hearing aids) that, at the investigator’s discretion, interferes with their ability to perform study assessments.5 |
|
Patients who have been diagnosed with arthritis or other musculoskeletal disorders affecting joints, muscles, ligaments, and/or nerves that by themselves affect the patient’s mobility and, at the investigator’s discretion, interfere with their ability to perform study assessments.5 |
fSARA is a modified version of SARA that was derived from FDA guidance to evaluate tangible changes in neurological symptoms and functioning1,2
The SARA is a highly reliable and consistent assessment tool that was validated to measure neurological changes.6
Functional SARA (fSARA) comprises functional assessments of 4 domains (derived from the 8 domains included in SARA), each rated from 0 to 4 points. Total fSARA score ranges from 0 to 16, with 0 representing the best neurological status and 16 the worst.1
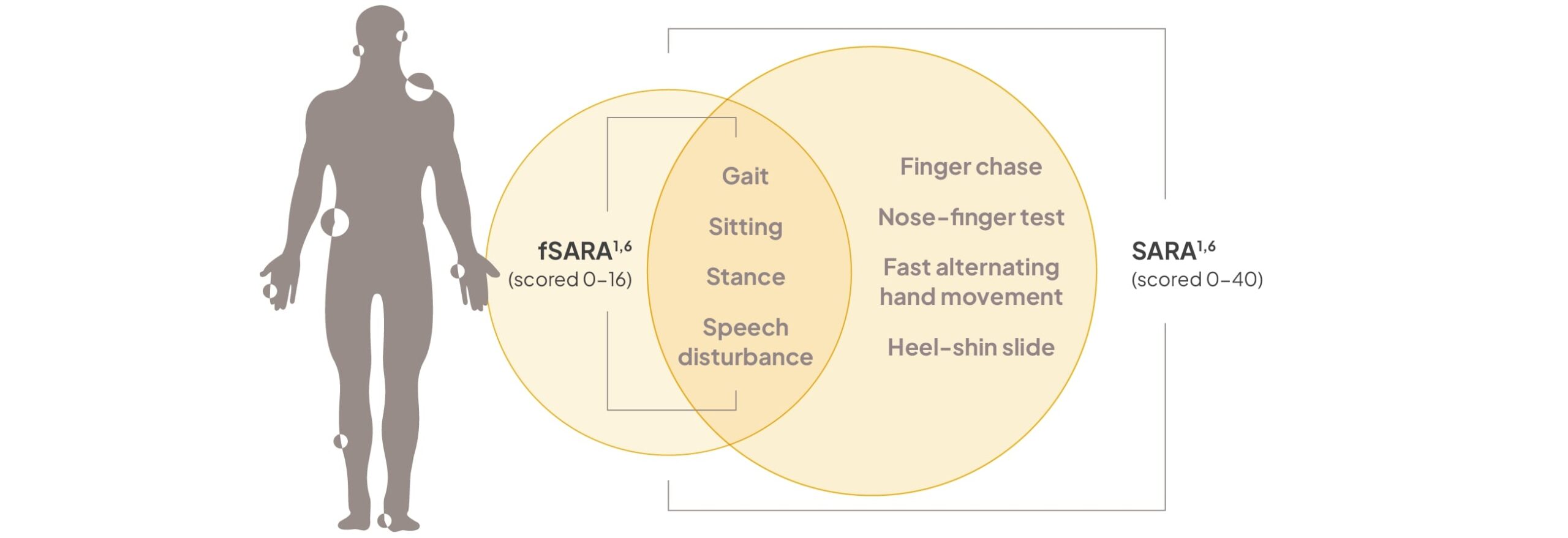
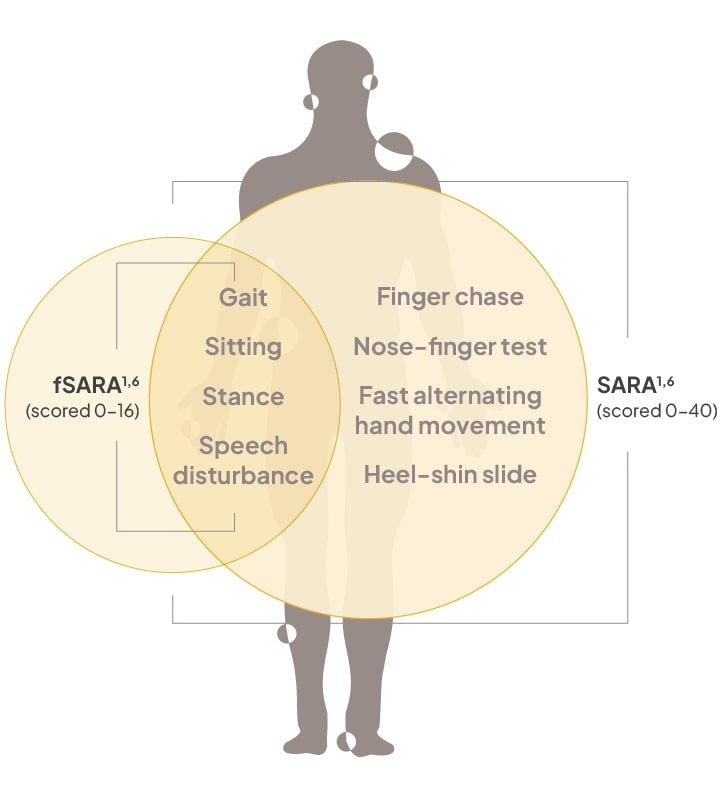
CGI-I, Clinical Global Impression of Improvement; mDRS, modified Disability Rating Scale; NPC-CSS, Niemann-Pick disease type C Clinical Severity Scale; SCAFI, Spinocerebellar Ataxia Functional Index.
Estimated treatment benefit of -0.4 points in mean total fSARA score with AQNEURSA vs placebo (95% CI: -0.7 to -0.2; two-sided P < 0.001)1


Consistent improvements in mean total fSARA score from baseline with AQNEURSA in both treatment sequences:


CI, confidence interval; SD, standard deviation.
Limitations: Subgroup analyses are exploratory and not confirmatory, as they were not prespecified in the study design. Results may have limited statistical power, and statistical significance should not be assumed.
These data are not included in the Prescribing Information for AQNEURSA. No conclusions regarding the benefits or risks can be established based solely on these data.
| Age group | |
|---|---|
| Pediatric (<18 years) | 23 (39%) |
| Adult (≥18 years) | 36 (61%) |
| Aged 4-9 years | 7 (12%) |
| Aged ≥10 years | 52 (88%) |
| SARA total score at baseline | |
|---|---|
| Median score >14.5 | 29 (49%) |
| Median score ≤14.5 | 30 (51%) |
| Miglustat use at visit 1, visit 2, or both | |
|---|---|
| Used miglustat | 50 (85%) |
| Did not use miglustat | 9 (15%) |
Limitations: These are interim results and should be interpreted with caution. Interim analyses are based on incomplete data and may be subject to change as the study progresses. Further analysis is needed to confirm these findings.
These data are not in the Prescribing Information for AQNEURSA. No conclusions regarding the benefits or risks can be established based solely on these data.
5-domain NPC-CSS assessments
Ambulation
Cognition
Speech
Swallowing
Fine motor skills
A 1-point or greater change in the 5-domain NPC-CSS score represents a clinically meaningful change, reflecting the gain (decrease in score) or loss (increase in score) of complex function, as well as disease modification (decrease in score) or disease progression (increase in score)7
Limitations: These are interim results and should be interpreted with caution. Interim analyses are based on incomplete data and may be subject to change as the study progresses. Further analysis is needed to confirm these findings.
These data are not in the Prescribing Information for AQNEURSA. No conclusions regarding the benefits or risks can be established based solely on these data.
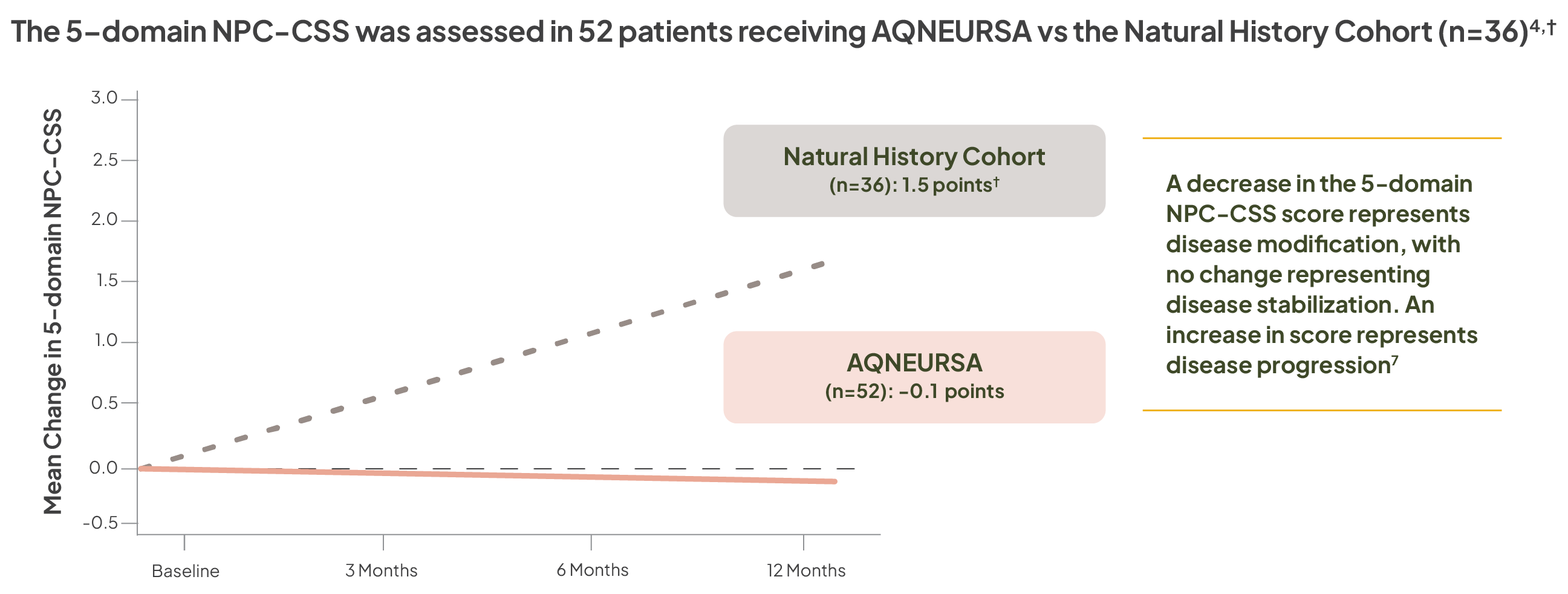
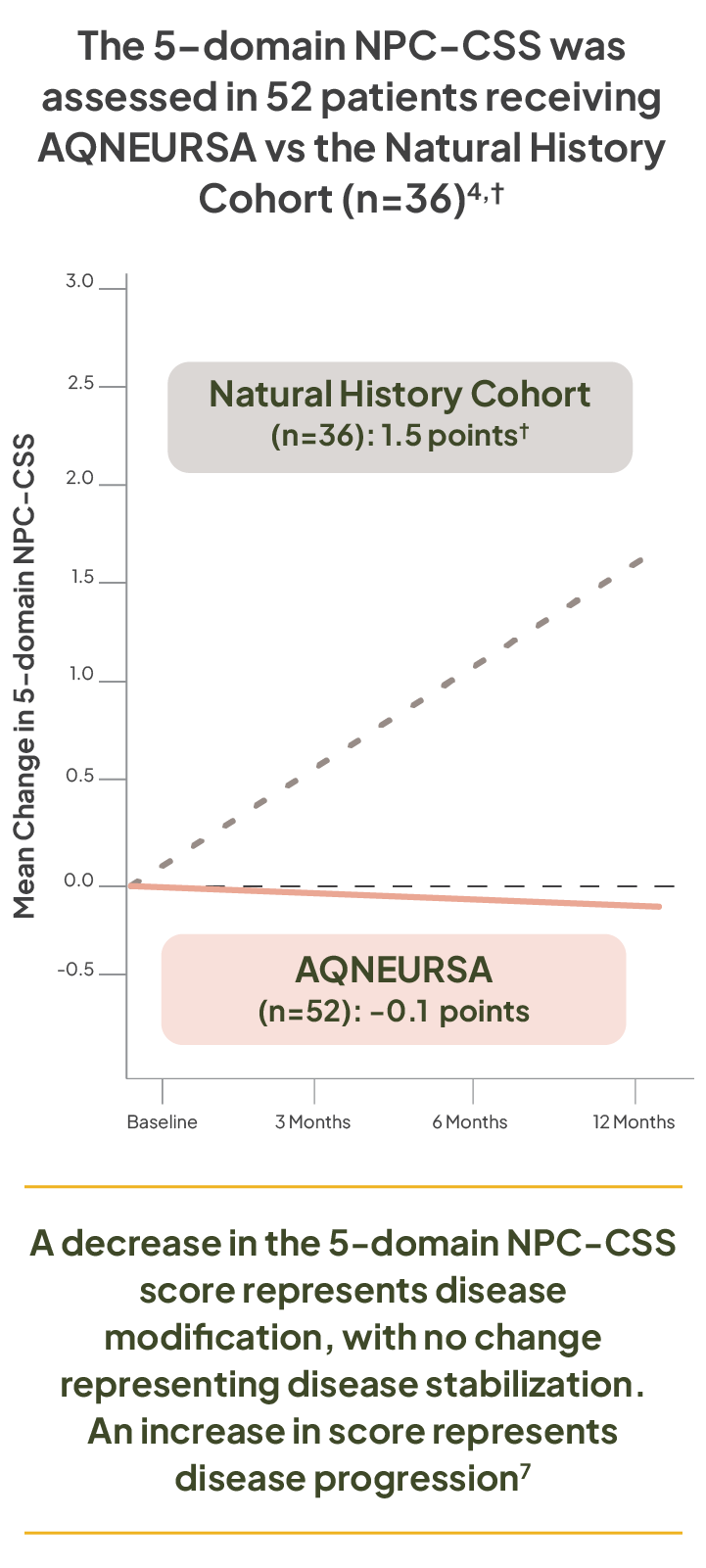
A decrease in the 5-domain NPC-CSS score represents disease modification, with no change representing disease stabilization. An increase in score represents disease progression7
| Mean change from baseline in total SARA score2,4 | |
|---|---|
|
After 12 weeks of treatment (pivotal trial, n=59) -2.0 ± 2.4 |
After 1 year of treatment (extension phase, n=52) -1.9 ± 2.9 |
†The Natural History Cohort is derived from the Prospective Natural History Study in NPC. Patients with NPC in the Natural History Cohort demonstrated an annualized linear progression of 1.5 points on the 5-domain NPC-CSS.4
References: 1. AQNEURSA. Prescribing information. IntraBio. 2. Bremova-Ertl T, Ramaswami U, Brands M, et al. Trial of N-acetyl-L-leucine in Niemann-Pick disease type C. N Engl J Med. 2024;390(5):421-431. doi:10.1056/NEJMoa2310151 3. Bremova-Ertl T, Ramaswami U, Brands M, et al. Trial of N-acetyl-L-leucine in Niemann-Pick disease type C. N Engl J Med. 2024;390(suppl 1):1-22. 4. Bremova-Ertl T, Rohrbach M, Ramaswami U, et al. Long-term findings of N-acetyl-L-leucine for Niemann-Pick disease type C. Presented at: 10th Congress of the European Academy of Neurology; June 29-July 2, 2024; Helsinki, Finland. 5. Data on file. IntraBio Inc. 6. Park J, Bremova-Ertl T, Brands M, et al. Assessment of the reliability, responsiveness, and meaningfulness of the Scale for the Assessment and Rating of Ataxia (SARA) for lysosomal storage disorders. J Neurol. 2024;271(10):6888-6902. doi:10.1007/s00415-024-12664-y 7.Yanjanin NM, Vélez JI, Gropman A, et al. Linear clinical progression, independent of age of onset, in Niemann-Pick disease, type C. Am J Med Genet B Neuropsychiatr Genet. 2010;153B(1):132-140. doi:10.1002/ajmg.b.30969